EtG, LMNOP, QRSTUV-What can EtG possibly tell us about whether or not someone was drinking….
Ethylglucuronide (EtG) testing across the United States is widely used by probation and parole offices to accuse a probationer/parolee of drinking. It can be a crippling accusation because oftentimes, the EtG tests are preformed far from clinical conditions and by folks who have not been sufficiently trained in the collection or interpretation of EtG testing.
EtG has been widely advanced as a “marker” of ETOH consumption since the 1950’s. However, widespread testing both in the criminal justice system and also in the workplace did not really begin until this millennium (the year 2000 and beyond) as the assaying for EtG became cheap, fast and easy to perform.
This goes back to a major theme of this blog. It is axiomatic that in science, you can only have either cheap, fast, easy or good. If you want cheap, you sacrifice typically specificity. If you want fast, you also typically sacrifice specificity. If you want easy, then you are never going to get good. To have a “good test” (meaning one that specific, precise, accurate (free of bias), sensitive, repeatable, reproducible, reliable, traceable, verifiable, valid, robust and as near the true result as scientifically possible) it will always take time, money and will be a process that requires training to faithfully execute the method.
Proponents of EtG testing claim that it is more sensitive and reliable indicator of both drinking and abstinence than was urine alcohol. However, that is not saying that much. [See our previous blog post “Urine ain’t worth piss”]
When we look at the attempts to validate the conclusion that EtG is a useful measure, we come to two basic assertions:
1. A positive EtG result is a valid way to prove that the person tested imbibed ETOH.
2. A negative EtG result is a valid way to prove that the person tested abstained from imbibing ETOH.
Both of these assertions by proponents of EtG testing fall short of the true scientific yard stick that is scientific validation (validation means documented proof that the process undertaken is suitable for its intended use and achieves the intended reported result correctly and uniquely.)
We must remember that according to the scientific method when properly applied, any experiment cannot prove the hypothesis. However, any one experiment can disprove a hypothesis. This is the idea of falsifying a hypothesis. So, if any experiment disproves your assertion or hypothesis, your hypothesis is rejected and there is no validation for that intended purpose. You have to go back to the proverbial drawing board.
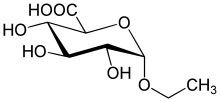
Let’s examine just assertion #1 in this blog: A positive EtG result is a valid way to prove that the person tested imbibed ETOH. Source: http://etg.weebly.com/ From their website:
- Incidental Exposure: Claims of “false positive EtG tests” from incidental exposure to alcohol began to occur. An online registry and listserve was developed for for those who claimed they’d been falsely accussed of drinking. The clamor of concern among this group rose rapidly and became extremely vigorous. This phenomenon of incidental exposure is very similar to the phenomenon of poppy seeds causing positive tests for morphine and is discussed in more depth in a separate section of this website.
- Elevated Concentration of Urine Artificially Increases EtG and EtS (and other analyte) levels: Because determination regarding the possibility of incidental exposure is in part determined by the level of EtG and/or EtS present in urine it is very important to consider the effects of concentration of reported levels. Few labs automatically adjust or correct EtG levels for concentration. Concentration is best estimated by level of creatinine in urine. The higher the creatinine the more concentrated the urine. Therefore, to adjust for the effect of concentration one can apply the same multiplier times EtG and/or EtS as would be used to reduce or increase creatinine to a given level. The level of 20, 50 or 100 can be used. For example, if urine creatinine is reported at 2,000ng/ml and the urine creatinine is 400mg/dl the U100EtG would be 500ng/ml because the mulitplier of 1/4th would be needed to reduce the creatinine to 100 and 1/4 times 2000 equals 500. Obviously correcting reported values to adjust for concentration can make significant changes at times and are important to consider, especially for EtG levels under 20,000ng/ml or so.
- Variation in amount of EtG produced for a given exposure to alcohol: Some individuals produce more EtG for a given exposure to alcohol than others. This is quit evident in exposure studies. Levels can vary dramatically between individuals, sometimes as much as 20-200 fold. This is important to keep in mind. There are likely individuals in the population that are “hyperproducers” of EtG and have much higher levels after incidental exposure.
- No Cutoff Known that Distinguishes Between Incidental Exposure and Drinking Alcohol: There is no known level of EtG or EtS which if exceeded indicates drinking. It is logical to assume that higher levels are more likely to be from drinking but there is no “bright line” that can distinguish between drinking and incidental exposure.
- Informed Consent: Individuals being tested with EtG and/or EtS should be thoroughly and carefully informed regarding items to avoid. A statement or contract addendum can be useful. (Download sample addendum used by some drug courts.)
- Topical Alcohol as a Source of Incidental Exposure: Volunteer exposure trials have demonstrated that alcohol-based handsanitizing gel causes positive EtG/EtS tests. A surprise finding has been that alcohol absorbed through inhalation of vapor, rather than through skin, is the chief cause of these positives .
- Stability and Synthesis of EtG: It was known that EtG could occasionally disappear (or be degraded) in urine stored at room temperature but not if frozen or heated. Researchers in Scandinavia further clarified this phenomenon when they reported that EtG (but not EtS) could be degraded in urine due to certain bacteria (explaining why heating or cooling samples resulted in less degradation). Furthermore, they then reported that in the presence of alcohol (fermented or added to urine) EtG could be synthesized by similar bacteria in-vitro if alcohol were present. This finding supported the likelihood that ethylsulfate (EtS), another minor metabolite of alcohol, may be a superior marker to EtG, in that it is more sensitive and specific.
- Reliability: Despite the possibility of incidental exposure most people who test positive for EtG actually did drink as the cause of the positive test. In one programs review, approximately 50% of individuals admitted drinking when supportively confronted. Another 40% subsequently admitted drinking at a later date. Those who initially deny drinking should receive more careful monitoring, testing, or other treatment, however, without other proof, they should not be presumed to have been drinking. SAMHSA advisory warning against over reliance on a positive EtG as sole proof of drinking.)
- Auto-Brewery Syndrome: The question of whether sugar can ferment to alcohol within the GI tract has been questioned. Recently, this question has achieved new importance as a study in Europe, soon to be published, has shown that volunteers consuming sugar and baker’s yeast orally produced elevated EtG/EtS levels.
As we can see just from examining assertion #1 of the proponents of the usefulness of EtG that it fails for various reasons. Therefore, scientifically there is no validated science that supports the hypothesis that a positive EtG result is a valid way to prove that the person tested actually truly imbibed ETOH. It makes me wonder why we use it at all. Perhaps it is the ease of use, but with that ease of use comes the cost of the truth. Truth should be in the courtroom, validated science should be in the courtroom, not “good enough for government work” when we are talking about people’s liberties and lives.



William C. Head says:
Excellent article. Better than the Pharmacologist that described it to our group on Sunday. Goes right into my trial notebook.
Justin J. McShane says:
Thank you Bubba. Just as you mentored me many years ago, it’s about giving back. I can’t be at every seminar and everywhere but thanks to the power of the internet, I can teach on demand to people in hopefully an understandable way to spread the word of both good and bad science.
Davides Constulopilos says:
A well written, fairly thourough article. However, you fail to mention the new SAMSHA advisory which establishes the cutoff for drinking vs. incedental exposure.
krm says:
Read your article. I was recently told by Dr Skipper that the 80 h ETG test is BS. WOuld you like to fill me in?
Example
person has 6-7 shot during a 3.5 hour period while eating food and drinking water, what would the ETG detection time be using a cutoff level of 500ng?
Thanks