Methamphetamine analysis comes down to the stereoselectivity capabilities in the crime laboratory.
Stereoselectivity is crucial in a crime laboratory. The ability of an analytical chemistry device to be specific to a type of a form of drug is key. Stereoselectivity is one of those key functions. Even the venerable workhorse of analytical chemistry in a crime laboratory-the Gas Chromatograph with Mass Spectrometry has a limitation. It’s limitation is in its ability to be stereoselective. As we blogged before, GC-MS is not perfect: The case study of methamphetamine there is a large issue in the ability of the GC-MS to distinguish between steroisomers of methamphetamine. From that post:
Both L-methamphetamine (levo-isomer referred to as the R-isomer) and D-methamphetamine (dextro-isomer referred to as the S-isomer) have the same chemical formula C10H15N. They have the same boiling point at 215 degrees Celsius at 760mmHG. They have the same molecular mass of 149.233 g/mol. They are stereoisomers.
And later on we make the following observation:
2.2.5.1 The L form has very little pharmacodynamic effect; whereas, the D will really make you have an interesting series of days.
2.2.5.2 The L-Form is known as Levomethamphetamine (other names include l-methamphetamine, levodesoxyephedrine, l-desoxyephedrine, levmetamfetamine).
2.2.5.3 The problem is that the L form is completely legal under certain circumstances, and is present in Vicks VapoRub® Inhaler which is over-the-counter for example (In fact, it has 50 mgs of the L for in a typical commercially available package). It is a common sympathomimetic vasoconstrictor. Per 21 CFR 1308.21 and 1308.22 the l-methamphetamine in VapoRub® Inhaler is non-narcotic. However, if one were to extract and collected from the inhalers and possessed with the intent to use, it would be impermissible (illegal) per the CFR.
Now you are asking yourself “So what does all of this have to do with my favorite show Breaking Bad?”
Do you remember this scene from Season 4 where Jesse Pinkman is brought down to the cartel to teach them how to do the “Heisenberg cook” of meth?
Live or die for Gus and company came down exclusively to the purity of the meth that was produced. Will the “Blue Sky” pass?
Well, thank goodness for the characters in the show the cartel folks are bad analytical chemists! to understand why I say that some background is needed.
To make a “better quality product,” Walter White shunned the hillbilly heroin approach of converting pseudoephedrine to methamphetamine. Using that Hillbilly Heroin method involving pseudoephedrine makes only the D form of meth that users want.
The P2P cook
There is a problem in the P2P cook. The P2P cook will make a racemic (science for “combined”) batch of both D and L form of methamphetamine. It will be a 50%/50% split of the form of the drug. Lucky for Jesse, the cartel didn’t know it’s analytical chemistry. They used a mass spec to arrive at the purity of the product that he made. The maximum that the MS could do is tell the purity of the racemic product and not whether or not you have pure D-methamphetamine (dextro-isomer referred to as the S-isomer) that the cartel wants. If the cartel really cared to make sure that all that was made by the P2P cook was in fact the D-form that users want, then they would need to use either a chiral column to chromatographically separate the D and the L forms, use a Circular dichroism spectroscopy unit or optical polarimetry unit, and/or a chiral detector.
In the real world, Walter White (aka Heisenberg) is using a “non-traditional” manufacturing process to make his methamphetamine. The writers of the show have stayed pretty close to the science of how this P2P cook is performed. The only liberty that the show producers have taken is in calling intermediary compound methylamine rare. In the real world, methylamine is not some sort of super rare and hard to synthesize matter. But hey, without it being supposedly rare how would we get that exciting train scene, right?
So, the P2P cook is simple from a chemist’s point of view.
Heisenberg starts his cook with a simple base product called phenylacetic acid (PAA) which is also known as α-toluic acid, benzeneacetic acid, alpha tolylic acid, 2-phenylacetic acid. It is a List 1 controlled chemical per the DEA found at 21 CFR 1310.02. Here it is:

He next wants to transform that chemical into PSP and into meth.
It is prepared by a scientific process called ketonization The ketonization of PAA and acetic acid over alumina occurs at 400-500 °C. This is why you see them adding aluminum chips into the big vat like so:

The aluminum oxide serves as a catalyst for the dehydration and decarboxylation (the ketonization), which is shown chemically as this:
C6H5CH2CO2H + CH3CO2H → C6H5CH2C(O)CH3 + CO2 + H2O
The result is the P2P.
P2P is C6H5CH2C(O)CH3. It’s full scientific name is phenyl-2-propanone (P2P) and is also known as Benzyl methyl ketone or Methyl benzyl ketone. Because it is known as a precursor of methamphetamine and amphetamine (phenyl ring with the carbon neck), it is currently a Schedule II drug in the United States. P2P or phenyl-2-propanone is this little guy:
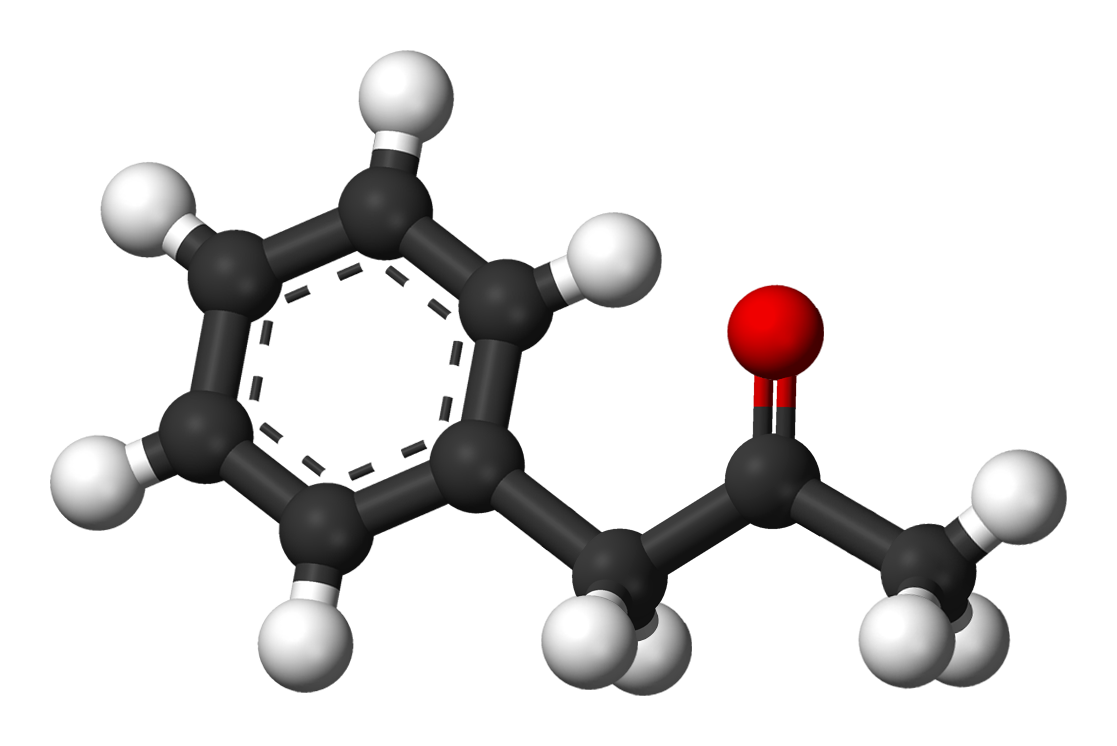
It is then reacted with the methylamine.
But you cannot stop there or you get a chemical that will not react pharmacodynamically. If you stop right here, you get an imine. An imine is a functional group or chemical compound containing a carbon–nitrogen double bond, with the nitrogen attached to a hydrogen atom (H) or an organic group.
Then good old fashiioned reductive amination (adding aluminum-mercury amalgam) is used to produce the final product known as N-methyl-1-phenylpropan-2-amine, the Schedule 1 Controlled Substance: meth which is pictured below:

The great mystery is the blue color of the methamphetamine that is made. Very pure methamphetamine is totally clear in color.

What’s the blue about? Only Heisenberg can be certain (Get it? Heisenberg uncertainty principle?)
Also, I know that I ruined the “romance” of the show by revealing that they make really good Vicks VapoRub too. Sorry.



Matthew Cowan says:
First of all I love breaking bad. But thru every season I couldn’t understand why they wouldn’t address the issue of racemic meth from p2p vs D meth from pseudo. And the methylamine, that was also very fantastic but it made good drama. When they first switched to the p2p cook and the blue color was introduced as pure meth I thought it was completely false and there would be a reason for it eventually. As the seasons progressed I was convinced the blue color was going to be revealed as a strategically placed impurity to brand his product. After the series ended I was really bummed that they didn’t sort out the racemic elephant in the room. But the blue color did end up serving the same purpose I predicted although never found to be put there purposely or strategically. W/o the blue color to brand Walt’s meth allot of the later stories couldn’t have been developed in the story.
Then I came across this Vice article written by an organic chemist and he offered up a reference to the possibility of enatiomeric resolution of methamphetamine isomers with tartaric acid. Great article.
“In a crystallographic resolution, a diasteromeric crystal or complex is formed between a chiral acid (like D-tartaric acid) and the compound so they can be separated. Unlike enantiomers, diastereomers have distinct physical properties that allow cooks to separate them using physical means like solubility. One chiral acid used to resolve methamphetamine is Di-p-toluoyl-tartaric acid ”
-http://motherboard.vice.com/blog/a-comprehensive-guide-to-the-clandestine-chemistry-of-breaking-bad
-13. Kozma D. Fogassy E. Optical Resolution of Methamphetamine by O,O’-Dibenzoyl-R,R-Tartaric Acid in Dichloroethane-Water-Methanol Solvent System. Synthetic Communications. 1999;29:4315-4319.